Storion lab
Заведующий
Федотов Станислав С
Публикаций
74
Цитирований
1 851
Индекс Хирша
22
Необходимо авторизоваться.
Overarched by the development of new energy storage technology our lab consists of three research groups, which are complementary to each other. Cathode, anode, electrolyte design and new chemical synthesis by Stanislav Fedotov Design of electrode/electrolyte interfaces and electrochemical study by Victoria Nikitina Computational design of battery materials and atomic level understanding of electrode/electrolyte interface by Dmitry Aksyonov
- Молекулярная динамика и квантово-химические расчеты
- Электрохимические исследования, в т.ч. operando дифракционные и спектроскопические эксперименты
- Различные виды неорганических синтезов: гидро(сольво)термальный, золь-гель, твердофазный
Всего публикаций
48
Всего цитирований
1759
Цитирований на публикацию
36.65
Среднее число публикаций в год
4
Годы публикаций
2013-2024 (12 лет)
h-index
22
i10-index
37
m-index
1.83
o-index
71
g-index
41
w-index
7
Описание метрик
h-index
Учёный имеет индекс h, если h из его N статей цитируются как минимум h раз каждая, в то время как оставшиеся (N - h) статей цитируются не более чем h раз каждая.
i10-index
Число статей автора, получивших не менее 10 ссылок каждая.
m-index
m-индекс ученого численно равен отношению его h-индекса к количеству лет, прошедших с момента первой публикации.
o-index
Среднее геометрическое h-индекса и числа цитирований наиболее цитируемой статьи ученого.
g-index
Для данного множества статей, отсортированного в порядке убывания количества цитирований, которые получили эти статьи, g-индекс это наибольшее число, такое что g самых цитируемых статей получили (суммарно) не менее g2 цитирований.
w-index
Если w статей ученого имеют не менее 10w цитирований каждая и другие статьи меньше, чем 10(w+1) цитирований, то w-индекс исследователя равен w.
Топ-100
Области наук
Журналы
Цитирующие журналы
Цитируемые журналы
Издатели
|
2
4
6
8
10
12
14
16
|
|
|
Elsevier
16 публикаций, 33.33%
|
|
|
American Chemical Society (ACS)
11 публикаций, 22.92%
|
|
|
Optica Publishing Group
4 публикации, 8.33%
|
|
|
Springer Nature
3 публикации, 6.25%
|
|
|
Royal Society of Chemistry (RSC)
3 публикации, 6.25%
|
|
|
The Electrochemical Society
3 публикации, 6.25%
|
|
|
MDPI
2 публикации, 4.17%
|
|
|
Wiley
1 публикация, 2.08%
|
|
|
Taylor & Francis
1 публикация, 2.08%
|
|
|
International Union of Crystallography (IUCr)
1 публикация, 2.08%
|
|
|
American Physical Society (APS)
1 публикация, 2.08%
|
|
|
Beilstein-Institut
1 публикация, 2.08%
|
|
|
OOO Zhurnal "Mendeleevskie Soobshcheniya"
1 публикация, 2.08%
|
|
|
2
4
6
8
10
12
14
16
|
Организации из публикаций
Страны из публикаций
|
5
10
15
20
25
30
35
40
45
50
|
|
|
Россия
|
Россия, 46, 95.83%
Россия
46 публикаций, 95.83%
|
|
Страна не определена
|
Страна не определена, 5, 10.42%
Страна не определена
5 публикаций, 10.42%
|
|
Германия
|
Германия, 4, 8.33%
Германия
4 публикации, 8.33%
|
|
США
|
США, 3, 6.25%
США
3 публикации, 6.25%
|
|
Китай
|
Китай, 3, 6.25%
Китай
3 публикации, 6.25%
|
|
Бельгия
|
Бельгия, 3, 6.25%
Бельгия
3 публикации, 6.25%
|
|
Италия
|
Италия, 3, 6.25%
Италия
3 публикации, 6.25%
|
|
Франция
|
Франция, 2, 4.17%
Франция
2 публикации, 4.17%
|
|
Украина
|
Украина, 1, 2.08%
Украина
1 публикация, 2.08%
|
|
Великобритания
|
Великобритания, 1, 2.08%
Великобритания
1 публикация, 2.08%
|
|
Испания
|
Испания, 1, 2.08%
Испания
1 публикация, 2.08%
|
|
Швеция
|
Швеция, 1, 2.08%
Швеция
1 публикация, 2.08%
|
|
5
10
15
20
25
30
35
40
45
50
|
Цитирующие организации
Цитирующие страны
- Мы не учитываем публикации, у которых нет DOI.
- Статистика пересчитывается раз в сутки.
Направления исследований
Development of computational framework SIMAN for high-throughput DFT calculations
+
https://github.com/dimonaks/siman
Understanding cation migration barriers in oxide and phosphate based cathode materials with DFT calculations
+
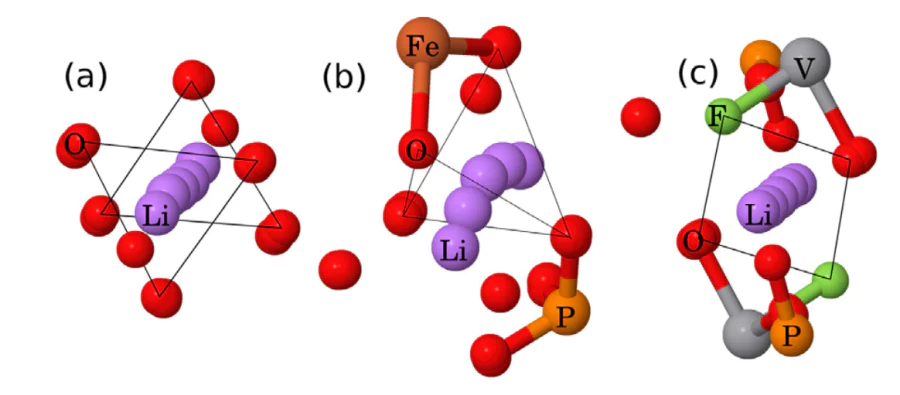
Computational Materials Science 154 (2018) 449–458
Advanced study of defects in electrodes for metal-ion batteries
+
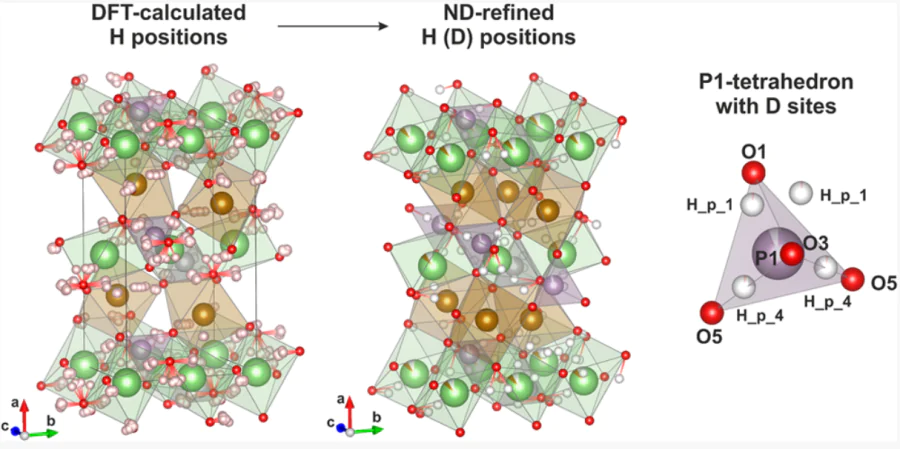
Inorg. Chem. 2021, 60, 5497−5506
Computational design of solid/solid and solid/liquid interfaces for metal-ion batteries
+
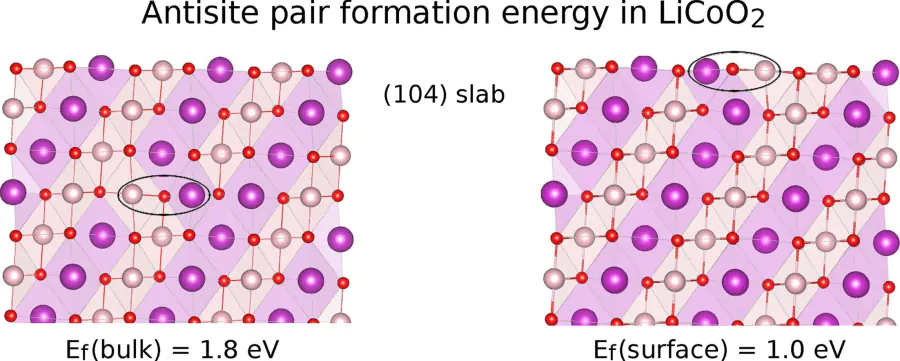
Computational design of solid/solid and solid/liquid interfaces for metal-ion batteries
Electrochemical CO2-to-fuels conversion
+

Electrochemical CO2 conversion to fuels and chemical feedstock is an attractive route to reduce carbon dioxide emissions to prevent catastrophic climate changes. We are focused on the design of non-precious metal-based electrocatalysts for CO2 conversion to synthesis gas, formate and alcohols and mechanistic studies of the electrochemical CO2 reduction pathways. We use electrodeposition to create high surface area bimetallic electrocatalysts based on Cu, Sn, Zn to convert greenhouse gas into valuable products.
(Photo)electrochemical hydrogen production
+
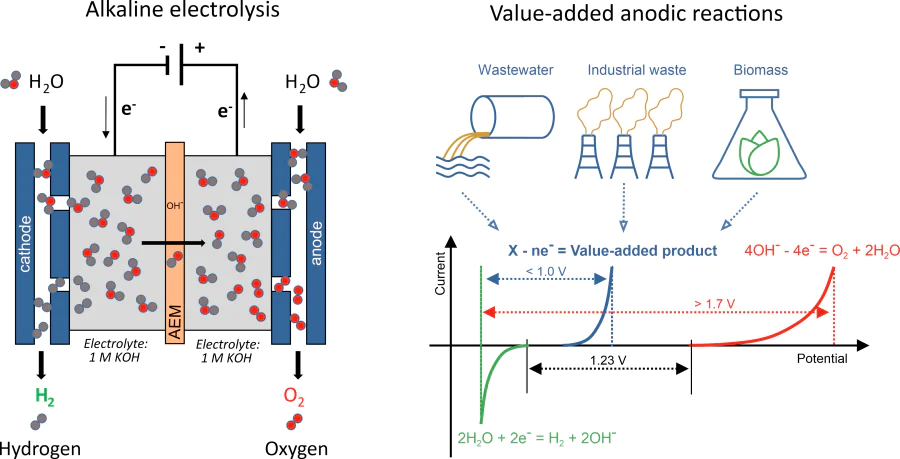
Green hydrogen, produced by electrolysis of aqueous solutions using renewable/sustainable energy sources, is a very much desired energy carrier, which could reduce our dependence on fossil fuels and decarbonize the industry. To date, 96% of hydrogen gas is still obtained from steam reforming and coal gasification, which leads to greenhouse gases emissions. Hydrogen produced by either acidic or alkaline electrolysis is still too expensive. The minimal voltage to break water molecules into hydrogen and oxygen is 1.23 V, while reaching industrially relevant current densities requires much higher voltages (more than 1.7 V). The cost of electricity thus plays a decisive role in the electrochemical hydrogen production. There is much more to be done to increase the chances for success of hydrogen-based economy. New electrocatalysts based on cheap and earth-abundant elements should be developed (currently most active electrocatalysts are based on Pt and Ir metals). Electrolyzer design must be improved to provide the necessary durability and cost-effectiveness. Using pure deionized water for electrolysis is a problem by itself, as it invokes the necessity of a pre-purification step. In our group, we focus on the following strategies to design efficient (photo)electrolyzers.
Low temperature ion insertion-based batteries
+

Li-ion batteries degrade quickly at subzero temperatures. To understand the origin of the battery performance deterioration we conduct multifaceted studies of all the factors, which are responsible for the changes in battery kinetics in the extended temperature range: changes in the activation barriers for ion intercalation, changes in the properties of the electrolytes and evolution of the electrode/electrolyte interphases. The ultimate goal is to design temperature-tolerant and safe battery chemistries, which deliver energy in the extended temperature interval (-100 - +100 °C). We focus on both non-aqueous Li-ion and Na-ion chemistries and aqueous rechargeable batteries (proton insertion batteries). Our current projects are: 1. Design of novel low-temperature electrolytes for Li-ion and Na-ion batteries to enable efficient operation in the extended temperature range. 2. Elucidating ion intercalation mechanisms using advanced electrochemistry and spectroscopy to derive information on the relevent rate-limiting steps. 3. Design of aqueous rechargeable proton-based batteries for ultra-high rate charge storage in the extended temperature range.
Публикации и патенты
Найдено
Ничего не найдено, попробуйте изменить настройки фильтра.
Адрес лаборатории
Москва, Большой бульвар, 30, стр. 1
Необходимо авторизоваться.