Лаборатория Полисераазотистых Гетероциклов
Публикаций
264
Цитирований
3 864
Индекс Хирша
31
Необходимо авторизоваться.
Синтез и химические свойства новых полисера- и сераазотистых гетероциклических соединений, которые могут быть ароматическими и антиароматическими, а также системами с расширенным сопряжением.
- Тонкий органический синтез
- ЯМР-спектроскопия
- Спектрофотометрия
- Рентгеноструктурный анализ
Олег Ракитин
Заведующий
Лидия Константинова
Ведущий научный сотрудник
Владимир Огурцов
Старший научный сотрудник
Екатерина Князева
Старший научный сотрудник
Максим Михайлов
Ведущий Инженер
Олег Большаков
Ведущий Инженер

Никита Гудим
Научный сотрудник
Наталья Обручникова
Младший научный сотрудник
Светлана Воронцова
Младший научный сотрудник
Михаил Толмачев
Аспирант
Ольга Устименко
Студент
Герман Чхетиани
Студент
Александр Постников
Студент
Клим Погорянский
Студент
Артем Чадин
Студент
Тимофей Кудряшев
Студент
Дарья Алехина
Студент
Карим Гайсин
Студент

Тимофей Кудряшев
Студент
Егор Котов
Студент
Всего публикаций
225
Всего цитирований
3277
Цитирований на публикацию
14.56
Среднее число публикаций в год
5.49
Годы публикаций
1983-2023 (41 год)
h-index
30
i10-index
109
m-index
0.73
o-index
68
g-index
41
w-index
5
Описание метрик
h-index
Учёный имеет индекс h, если h из его N статей цитируются как минимум h раз каждая, в то время как оставшиеся (N - h) статей цитируются не более чем h раз каждая.
i10-index
Число статей автора, получивших не менее 10 ссылок каждая.
m-index
m-индекс ученого численно равен отношению его h-индекса к количеству лет, прошедших с момента первой публикации.
o-index
Среднее геометрическое h-индекса и числа цитирований наиболее цитируемой статьи ученого.
g-index
Для данного множества статей, отсортированного в порядке убывания количества цитирований, которые получили эти статьи, g-индекс это наибольшее число, такое что g самых цитируемых статей получили (суммарно) не менее g2 цитирований.
w-index
Если w статей ученого имеют не менее 10w цитирований каждая и другие статьи меньше, чем 10(w+1) цитирований, то w-индекс исследователя равен w.
Топ-100
Области наук
Журналы
Цитирующие журналы
Издатели
|
10
20
30
40
50
60
|
|
|
Springer Nature
53 публикации, 23.56%
|
|
|
MDPI
46 публикаций, 20.44%
|
|
|
Elsevier
31 публикация, 13.78%
|
|
|
OOO Zhurnal "Mendeleevskie Soobshcheniya"
23 публикации, 10.22%
|
|
|
Royal Society of Chemistry (RSC)
21 публикация, 9.33%
|
|
|
Wiley
12 публикаций, 5.33%
|
|
|
American Chemical Society (ACS)
12 публикаций, 5.33%
|
|
|
Autonomous Non-profit Organization Editorial Board of the journal Uspekhi Khimii
8 публикаций, 3.56%
|
|
|
Pleiades Publishing
5 публикаций, 2.22%
|
|
|
Taylor & Francis
4 публикации, 1.78%
|
|
|
Georg Thieme Verlag KG
4 публикации, 1.78%
|
|
|
ARKAT USA, Inc.
2 публикации, 0.89%
|
|
|
International Union of Crystallography (IUCr)
1 публикация, 0.44%
|
|
|
Beilstein-Institut
1 публикация, 0.44%
|
|
|
Hindawi Limited
1 публикация, 0.44%
|
|
|
10
20
30
40
50
60
|
Организации из публикаций
Страны из публикаций
|
20
40
60
80
100
120
140
160
180
200
|
|
|
Россия
|
Россия, 195, 86.67%
Россия
195 публикаций, 86.67%
|
|
Страна не определена
|
Страна не определена, 35, 15.56%
Страна не определена
35 публикаций, 15.56%
|
|
Великобритания
|
Великобритания, 35, 15.56%
Великобритания
35 публикаций, 15.56%
|
|
Испания
|
Испания, 9, 4%
Испания
9 публикаций, 4%
|
|
США
|
США, 5, 2.22%
США
5 публикаций, 2.22%
|
|
Финляндия
|
Финляндия, 5, 2.22%
Финляндия
5 публикаций, 2.22%
|
|
Швейцария
|
Швейцария, 5, 2.22%
Швейцария
5 публикаций, 2.22%
|
|
Германия
|
Германия, 3, 1.33%
Германия
3 публикации, 1.33%
|
|
Китай
|
Китай, 3, 1.33%
Китай
3 публикации, 1.33%
|
|
Кипр
|
Кипр, 3, 1.33%
Кипр
3 публикации, 1.33%
|
|
Франция
|
Франция, 1, 0.44%
Франция
1 публикация, 0.44%
|
|
Бразилия
|
Бразилия, 1, 0.44%
Бразилия
1 публикация, 0.44%
|
|
Италия
|
Италия, 1, 0.44%
Италия
1 публикация, 0.44%
|
|
Норвегия
|
Норвегия, 1, 0.44%
Норвегия
1 публикация, 0.44%
|
|
Япония
|
Япония, 1, 0.44%
Япония
1 публикация, 0.44%
|
|
СССР
|
СССР, 1, 0.44%
СССР
1 публикация, 0.44%
|
|
20
40
60
80
100
120
140
160
180
200
|
Цитирующие организации
Цитирующие страны
- Мы не учитываем публикации, у которых нет DOI.
- Статистика пересчитывается раз в сутки.
Направления исследований
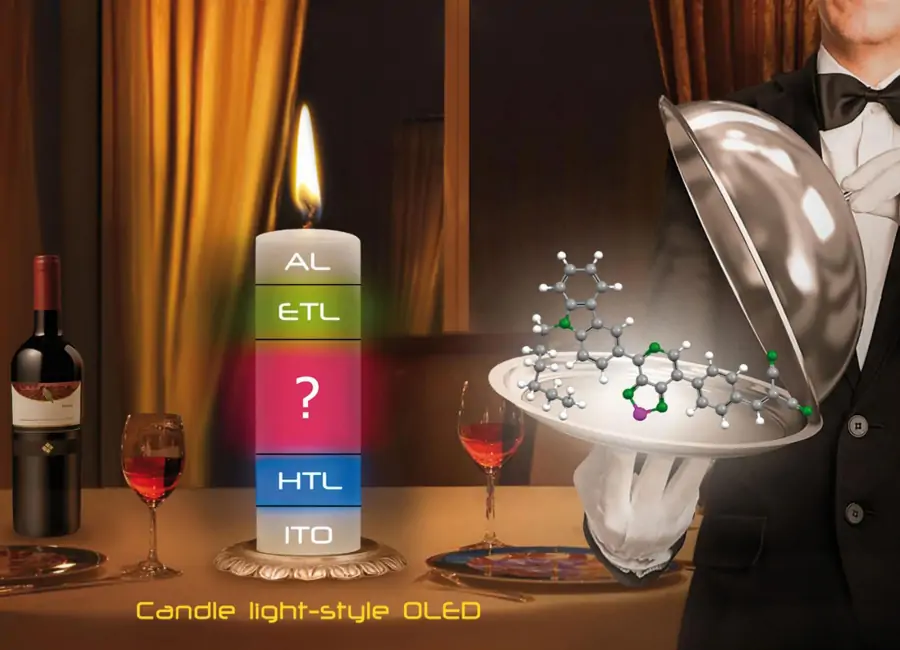
Получение активных компонентов органических светоизлучающих диодов со световыми характеристиками свечей (Candle Light-Style Organic Light Emitting Diode)
+

Поиск оптимальных структур и направленный синтез красителей на основе халькогеназот содержащих гетероцилов для создания безопасных с физиологической точки зрения светодиодов. Такие светодиоды, благодаря инновационной конструкции и сочетанию новых специально синтезированных люминофоров, практически не имеют вредного излучения в синей области спектра, а цветовые характеристики их излучения подобны световым характеристикам пламени обычных свечей. Преимуществом таких устройств является также отсутствие в их составе редких и благородных металлов, таких как иридий и платина (основы современных промышленных люминофоров для OLED устройств), и относительная простота конструкции в целом.
Новые подходы к полисераазотсодержащим гетероциклам
+
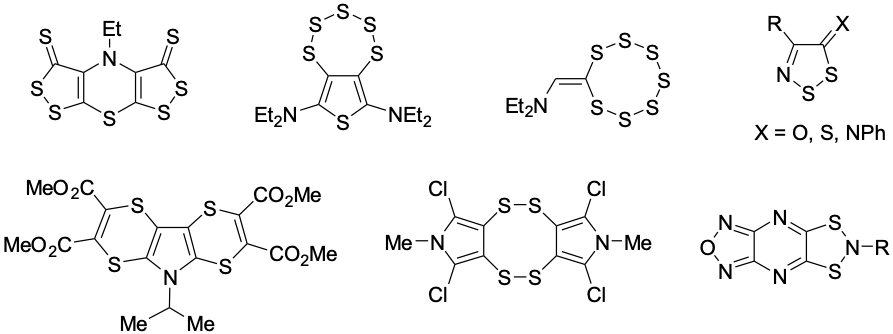
Исследуются новые подходы к синтезу полисераазотсодержащих гетероциклов, содержащих несколько атомов серы и атом азота на основе реакции комплексов монохлорида серы и азотсодержащих оснований. На данный момент получен ряд новых гетероциклических систем, в том числе 1,2- и 1,4-тиазины, тиофены, моноциклические и конденсированные 1,2-дитиолы и 1,2,3-дитиазолы, 1,2,3,4,5-пентатиепины, 1,2,3,4,5,6,7-гептатиоканы и др. Синтезированные нами полисерасодержащие гетероциклические соединения (1,2-дитиолы, 1,2,3,4,5-пентатиепины и 1,2,3-дитиазолы) показывают высокую противораковую, противомикробную и противогрибковую виды активности.Синтезированные 1,2,3,4,5-пентатиепины были изучены в качестве катодного материала для аккумуляторных батарей; конденсированные 1,4-дитиины предложены в качестве компонентов многоцелевого назначения для фоторецепторов.
Новые серии органических сенсибилизаторов на основе халькогеназотсодержащих гетероциклов
+
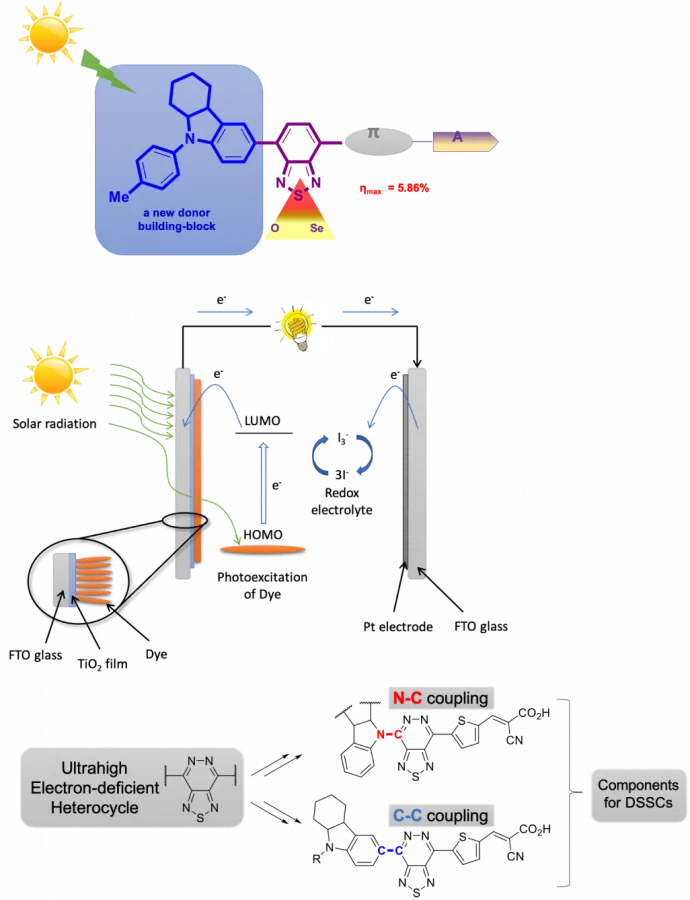
Поиск новых эффективных органических компонентов для сенсибилизированных красителем солнечных батарей (Dye Sensitized Solar Cells, DSSCs) на основе стратегии D-A-π-А, заключающаяся во введении между донором (D) и π-мостиком дополнительной акцепторной группы (А), в роли которой выступают халькогеназотсодержащие гетероциклы. Разработка органических сенсибилизаторов более сложного дизайна для создания более эффективных DSSCs.
Общий механизм действия полисернистых гетероциклов против вируса иммунодефицита кошек
+
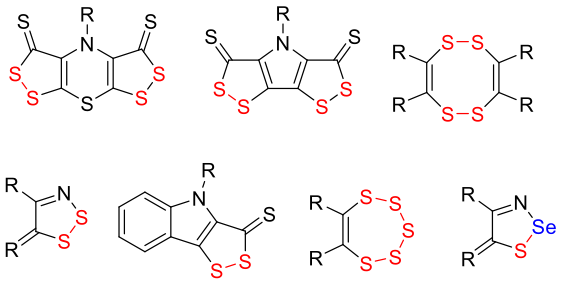
Разрабатываются методы синтеза полисернистых гетероциклов, которые обладают общим механизмом действия и высокой селективностью против вируса иммунодефицита кошек, являющийся ближайшим аналогом вируса иммунодефицита человека. Ранее уже было установлено, что соединения разной гетероциклической структуры: бис(дитиоло)тиазины, бис(дитиоло)пирролы, тетратиоцины, дитиазолы, дитиолоиндолы, пентатиепины и тиаселеназолы – являются перспективными антиспидовыми соединениями, что представляет значительную ценность для развития не только химической, но и биологической наук.
Публикации и патенты
Найдено
Ничего не найдено, попробуйте изменить настройки фильтра.
Чмовж Т.Н., Князева Е.А., Ракитин О.А.
RU2668978C1,
2018
В. Н. Коршунов, Т. Н. Чмовж, Е. А. Князева, И. В. Тайдаков, Л. В. Михальченко, Е. А. Вараксина, Р. Р. Сайфутяров, И. Х. Аветисов, О. А. Ракитин
RU2729424C1,
2020
Адрес лаборатории
ИОХ РАН, 311 ГК, 505 ЛК, 506 ЛК, 509 ЛК, 201 КМУ
Необходимо авторизоваться.